Moderna Vaccine California
Moderna vaccine has poisonous SM-102 chemical in it. Kirkland did get the vaccine.
Riverside County Albertsons Stores Temporarily Stop Accepting Appointments For Covid 19 Vaccinations Kesq
In that time period more than 272000 people received the vaccine.

Moderna vaccine california. Moderas vaccine is authorized for emergency use in those 18 and older. The Moderna COVID19 vaccine pINN. Her friend recovered but five months after her second Moderna shot Kirkland remains in constant pain.
180 million Americans fully vaccinated Today during a White House briefing Jeff Zients the COVID-19 repose coordinator said 180 million Americans were fully vaccinated against COVID-19. Last month Pfizer and BioNTech announced they had started the approval process. Of anaphylaxis in response to any COVD-19 vaccine not just Moderna.
Pfizer and BioNTechs vaccine is authorized by the Food and Drug Administration for emergency use in people ages 16 and older. The Defender is experiencing censorship on many social channels. Moderna submitted its Biologics License Application to the FDA about one month after Pfizers submission meaning that full approval of its vaccine.
After seeing what a friend who got the virus went through she got both doses of the Moderna vaccine. Anthony Fauci of The National Institutes of Health say that the Moderna vaccines mRNA components would have a similar success rate to. FDA authorized the emergency use of the Moderna COVID-19 Vaccine in individuals 18 years of age or older.
It is authorized for use in people aged twelve years and older in some. Medical experts such as Dr. About the Moderna COVID-19 Vaccine.
California pauses use of Moderna vaccine batch over allergic reactions California health officials are asking to pause the use of a huge batch of Modernas coronavirus vaccine due to its higher-than-usual number of possible allergic reactions. Moderna says its COVID-19 vaccine candidate is 945 effective. Pfizer says its vaccine candidate co-developed with BioNTech is 95 effective.
SAN FRANCISCO KGO -- Over the past few weeks California has rapidly expanded its vaccine eligibility meaning millions of people are getting a. The Moderna COVID-19 Vaccine is an mRNA vaccine against COVID-19 encoding for a prefusion stabilized form of the Spike S protein. Moderna filed for final approval of its vaccine on June 1 and expects to complete its submission in August.
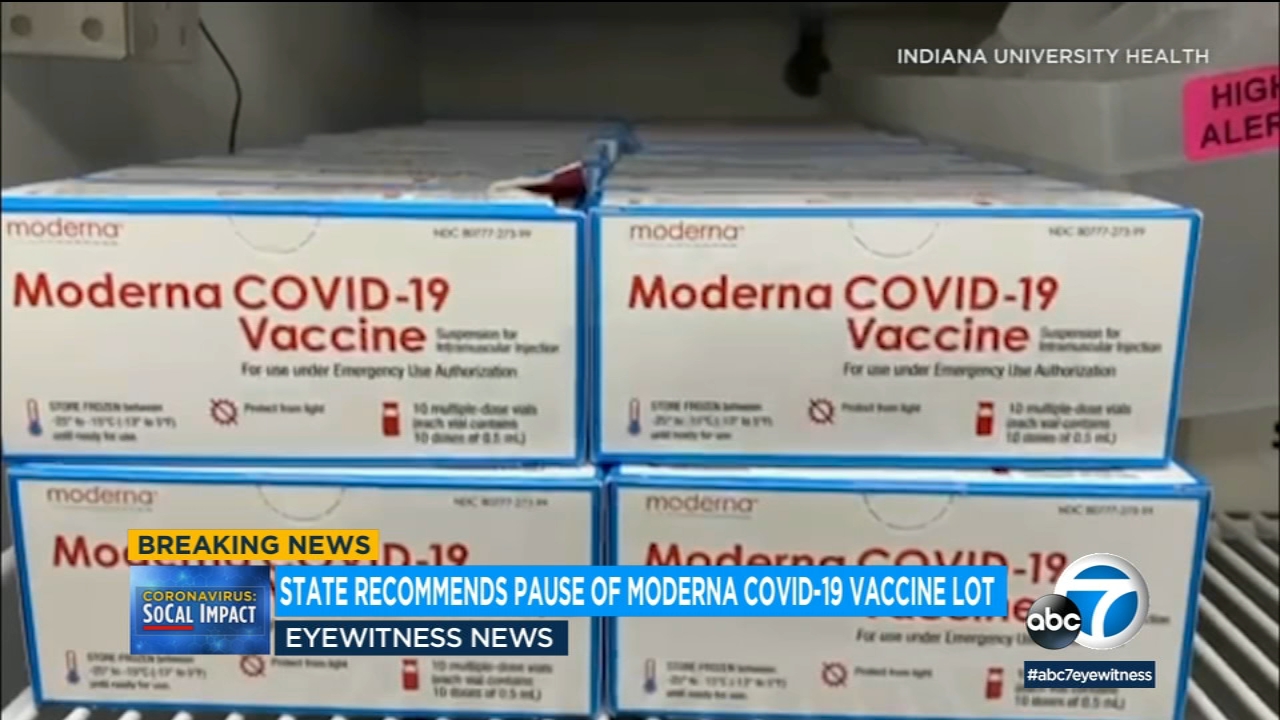
The California Department of Public Health has opened an investigation into one lot of a COVID vaccine from Moderna after it possibly caused several severe allergic reactions. Though the headline crows about Hopeful Results in the Moderna COVID vaccine trials by the end of the paper we learn that 21 of people receiving the vaccine experienced a serious event. Moderna said its COVID-19 vaccine remained 93 effective through six months after the second shot.
19 there have been six cases in the US. To authorize a booster dose this month. Additionally the vaccine seems to have broad efficacy with success rates consistent across various age race ethnicity and gender groups.
More than 60 of adults have now received at least one dose of a coronavirus vaccine and as the CDC has issued new guidance on mask. A health care worker fills a syringe with Moderna COVID-19 vaccine as California opens up vaccine eligibility to any residents 16 years and older during the. The lot which.
Moderna Pediatric COVID-19 Vaccine Study Survey Who We Are Rady Childrens Hospital-San Diego is a 505-bed pediatric care facility providing the largest source of comprehensive pediatric medical services in San Diego southern Riverside and Imperial counties. Moderna will begin studying its COVID vaccine in pregnant women according to a posting on ClinicalTrialsgovThe observational study expected to begin July 22 will enroll about 1000 females over age 18 who will be. Again recipients of the Moderna vaccine followed by Pfizer then JJ had higher anti-spike Immunoglobulin G levels a sign of a stronger antibody response.
Moderna is the second drugmaker in the US. On December 18 2020 the US. California has received about 330000 doses from the Moderna lot in question about 10 of all the vaccine both Modernas and Pfizers that have been distributed across the Golden State.
Be sure to stay in touch with the news that matters by subscribing to our top news of the day. As of Dec. See Media Release here.
Elasomeran codenamed mRNA-1273 and sold under the brand name Spikevax is a COVID-19 vaccine developed by American company Moderna the United States National Institute of Allergy and Infectious Diseases NIAID and the Biomedical Advanced Research and Development Authority BARDA. Moderna a Massachusetts-based vaccine developer partnered with the National Institutes of Health to develop and test a coronavirus vaccine known as mRNA-1273A clinical trial demonstrated that. To begin applying for full FDA approval for its COVID-19 vaccine.
COVID in California. Pfizer is also expected to ask the FDA. Health officials are investigating reports that Moderna Incs COVID-19 vaccine may be linked to a higher risk of a rare heart condition in younger adults than previously thought the Washington Post reported late on Thursday citing people familiar with the review.
Modernas vaccine has been shown in trials to be very effectivepreventing COVID-19 in 941 of the people enrolled in the Phase 3 clinical trial. Modernas latest efficacy data hasnt been published in. The administration of over 330000 doses of Modernas covid vaccine were halted in California as health officials investigate higher than usual reactions that occurred at San Diego Countys.
Moderna vaccine could be twice as effective as Pfizer against delta study finds Aidin Vaziri Rita Beamish Dominic Fracassa Anna Buchmann Aug. Modernas COVID-19 vaccine is currently authorized under an Emergency Use Authorization EUA for adults 18 and up.

Moderna Says Early Data Shows Covid Vaccine Is 96 Effective In Teens

Here S What Healthcare Workers Do With Extra Covid 19 Vaccines At The End Of The Day Abc4 Utah
Fda Asks Moderna Pfizer To Add More Children To Covid 19 Vaccine Efficacy Studies
California Calls For Pausing Distribution Of Specific Lot Of Moderna Vaccine After Allergic Reactions Reported Abc7 New York
Trinity County Covid 19 Second Dose Of Moderna Vaccine Available

California Calls For Pausing Distribution Of Specific Lot Of Moderna Vaccine After Allergic Reactions Reported Abc7 New York
Moderna Vaccines On Hold Due To Allergic Reactions Found In Tulare Kings Kern Counties Kmph

Ca Public Health On Twitter The Ingredients In The Covid 19 Vaccines Are No Mystery Read What S In The Moderna Pfizer And Johnson Johnson Vaccines Https T Co Le318hxhgk

Drive Through Covid 19 Mass Vaccination Clinic Scheduled For Saturday In Salinas Monterey Herald

California Woman Arrested For Making Fake Vaccine Cards Justice Department Says

State Recommends Providers Pause Administration Of Specific Moderna Covid 19 Vaccine Lot Following Possible Allergic Reactions Yourcentralvalley Com

5 San Francisco Bay Area Counties Say Providers Received Moderna Vaccine Lot Paused For Allergic Reaction Investigation Abc7 San Francisco
Moderna Vaccine Protects Against Covid For Months Company Says Los Angeles Times

California Ok To Use Moderna Vaccine After Illness Reports Kpbs

Moderna Vaccine Protects Against Covid For Months Company Says Los Angeles Times

California Ag Don T Buy Or Print Fake Covid 19 Vaccination Cards

How The New One Dose Covid 19 Vaccine Compares To Pfizer And Moderna Daily News

Who Says Pregnant Women Should Not Get Moderna Vaccine The Times Of Israel
/cloudfront-us-east-2.images.arcpublishing.com/reuters/SU2QKHGSLVPEHIBIMJUA4ONZRA.jpg)
Moderna Says Its Covid 19 Vaccine Found Safe Effective In Teens Reuters


Post a Comment for "Moderna Vaccine California"